The Effects of Different Concentrations of (NH₄)₂SO₄ on Daphnia
Introduction:
Water pollution and contamination have harmful effects on both the organisms that live in and consume water. If the contamination was strong enough, it could do as much as kill 50% or more the population. Different levels of water toxicity could also do other harmful damage such as cause birth defects and problems, health complications and behavior changes (LennTech). Aquatic toxicity could also influence the food chain of an ecosystem, which could possibly lead to the endangerment or extinction of one or more species ("Sublethal Toxic Effects in a Simple Aquatic Food Chain", ScienceDirect). The effects of water toxicity are serious and greatly affect the environment (plant life) and aquatic (fish, crustaceans, algae, etc.) and terrestrial (mammals, reptiles, avians, insects, etc.) organisms ("Pesticides: Environmental Effects", EPA). Governmental programs such the Environmental Protection Agency (EPA) investigates upon these issues in order to find solutions to the problems and ways of preventions, such as regulation of wastes and release of toxic compounds into air and water. This is often accomplished by identifying the sources of the pollution so that further damage could be prevented. The pollutant can originate from "readily identifiable sources through which the pollutants enter the water", point sources, or from sources that are more difficult to identify and control, nonpoint sources. Point sources can range from "wastewater effluent pipes or sewage discharge points" while nonpoint sources are more widespread and can enter through "runoff, precipitation, and seepage" (LC50 Aquatic Toxicity, Carolina EcoKits).
The ways in which the EPA investigates environmental issues such as water pollution include the intended exposure of indicators (animal or plant species that can be used to infer conditions in specific habitats due to their sensitivity to changes of the conditions in their habitat) to different concentrations of pollutants, such as ammonium sulfate (NH₄)₂SO₄. This procedure serves to test the levels of toxicity (the degree to which toxic or poisonous substances could harm organisms) of ammonium sulfate in water and what specific concentration of the pollutant terminates half of the sample population of the Daphnia magna (Lethal Concentration 50/LC50). If Daphnia were to be exposed to different concentrations of (NH₄)₂SO₄, then half of the sample population would be terminated at 0.03% concentration of (NH₄)₂SO₄.
Methods and Materials:
In order to identify the effects of different levels of water toxicity on the environment, the indicator organism Daphnia magna was exposed to different concentrations of (NH₄)₂SO₄. This was done by setting up five sets of five cups filled with 70mL of ammonium sulfate solutions with different concentration. Each set had one control variable, the 0.0% concentration (just spring water) and four experimental variables, the 0.02%, 0.03%, 0.04%, and 0.05% concentrations (ammonium sulfate solutions). Once the cups were set accordingly, five Daphnia were placed into each cup. After the experiment was set aside for 24 hours, the amount of living Daphnia in each concentration was counted and the data was collected. The materials used in this procedure included a permanent marker, 5 plastic cups 5 pipets, 5 ammonium sulfate solutions in labeled containers (0.0%, 0.02%, 0.03%, 0.04%, and 0.05%), 5 labeled 100-mL graduated cylinders (each corresponding to one solution), and Daphnia magna. The 5 labeled 100-mL graduated cylinders were used to distribute the 5 ammonium sulfate solutions into the 5 plastic cups (labeled using the permanent marker) and the pipets were used to transfer the Daphnia magna into the 5 ammonium sulfate solutions.
There was a total of five groups per class for the one class. Each group had 25 Daphnia magna and 5 ammonium sulfate solutions. To be able to observe the effects of different levels of water toxicity, each group had a 0.0% concentration solution of ammonium sulfate as the control variable and 0.02%, 0.03%, 0.04%, and 0.05% concentrations as the experimental variable.
Water pollution and contamination have harmful effects on both the organisms that live in and consume water. If the contamination was strong enough, it could do as much as kill 50% or more the population. Different levels of water toxicity could also do other harmful damage such as cause birth defects and problems, health complications and behavior changes (LennTech). Aquatic toxicity could also influence the food chain of an ecosystem, which could possibly lead to the endangerment or extinction of one or more species ("Sublethal Toxic Effects in a Simple Aquatic Food Chain", ScienceDirect). The effects of water toxicity are serious and greatly affect the environment (plant life) and aquatic (fish, crustaceans, algae, etc.) and terrestrial (mammals, reptiles, avians, insects, etc.) organisms ("Pesticides: Environmental Effects", EPA). Governmental programs such the Environmental Protection Agency (EPA) investigates upon these issues in order to find solutions to the problems and ways of preventions, such as regulation of wastes and release of toxic compounds into air and water. This is often accomplished by identifying the sources of the pollution so that further damage could be prevented. The pollutant can originate from "readily identifiable sources through which the pollutants enter the water", point sources, or from sources that are more difficult to identify and control, nonpoint sources. Point sources can range from "wastewater effluent pipes or sewage discharge points" while nonpoint sources are more widespread and can enter through "runoff, precipitation, and seepage" (LC50 Aquatic Toxicity, Carolina EcoKits).
The ways in which the EPA investigates environmental issues such as water pollution include the intended exposure of indicators (animal or plant species that can be used to infer conditions in specific habitats due to their sensitivity to changes of the conditions in their habitat) to different concentrations of pollutants, such as ammonium sulfate (NH₄)₂SO₄. This procedure serves to test the levels of toxicity (the degree to which toxic or poisonous substances could harm organisms) of ammonium sulfate in water and what specific concentration of the pollutant terminates half of the sample population of the Daphnia magna (Lethal Concentration 50/LC50). If Daphnia were to be exposed to different concentrations of (NH₄)₂SO₄, then half of the sample population would be terminated at 0.03% concentration of (NH₄)₂SO₄.
Methods and Materials:
In order to identify the effects of different levels of water toxicity on the environment, the indicator organism Daphnia magna was exposed to different concentrations of (NH₄)₂SO₄. This was done by setting up five sets of five cups filled with 70mL of ammonium sulfate solutions with different concentration. Each set had one control variable, the 0.0% concentration (just spring water) and four experimental variables, the 0.02%, 0.03%, 0.04%, and 0.05% concentrations (ammonium sulfate solutions). Once the cups were set accordingly, five Daphnia were placed into each cup. After the experiment was set aside for 24 hours, the amount of living Daphnia in each concentration was counted and the data was collected. The materials used in this procedure included a permanent marker, 5 plastic cups 5 pipets, 5 ammonium sulfate solutions in labeled containers (0.0%, 0.02%, 0.03%, 0.04%, and 0.05%), 5 labeled 100-mL graduated cylinders (each corresponding to one solution), and Daphnia magna. The 5 labeled 100-mL graduated cylinders were used to distribute the 5 ammonium sulfate solutions into the 5 plastic cups (labeled using the permanent marker) and the pipets were used to transfer the Daphnia magna into the 5 ammonium sulfate solutions.
There was a total of five groups per class for the one class. Each group had 25 Daphnia magna and 5 ammonium sulfate solutions. To be able to observe the effects of different levels of water toxicity, each group had a 0.0% concentration solution of ammonium sulfate as the control variable and 0.02%, 0.03%, 0.04%, and 0.05% concentrations as the experimental variable.
Results:
Conclusion:
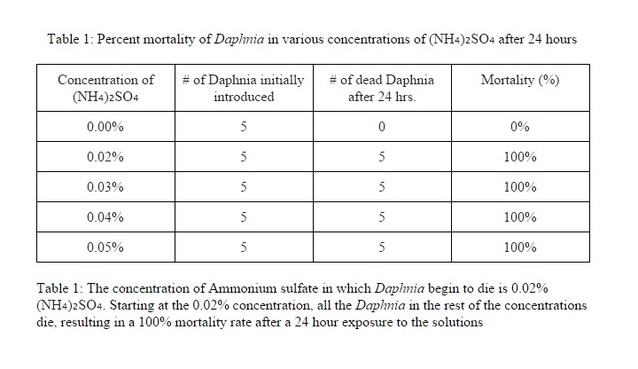
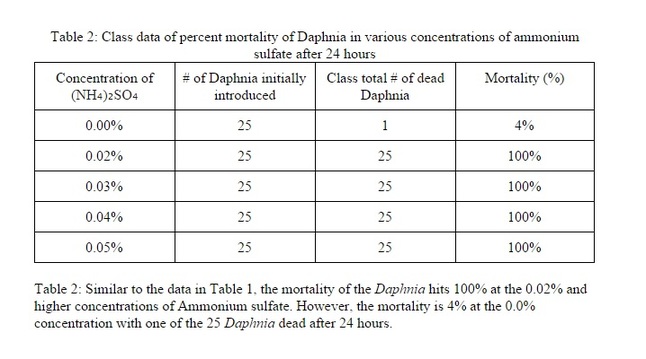
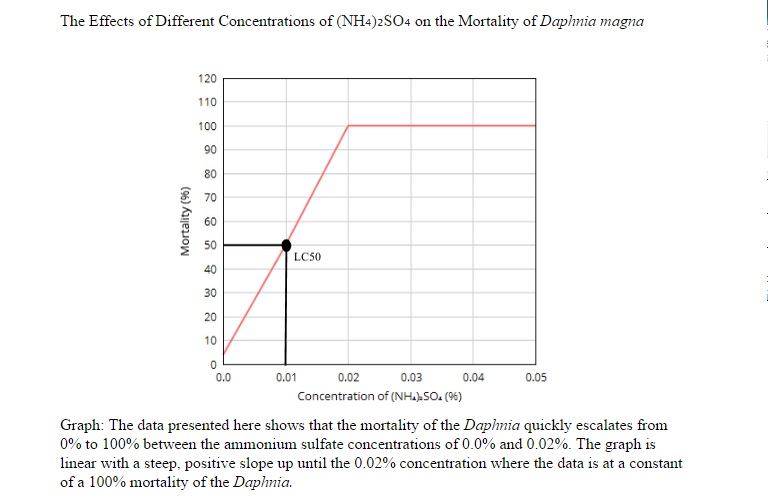
According to the results in Table 1 and Table 2, the Daphnia magna all die starting at the 0.02% concentration of (NH₄)₂SO₄ with the exception of only one death in the 0.0% concentration in the class data. The graph illustrates that the mortality of the Daphnia quickly rises from 0% to 100% in the concentrations between 0.0% and 0.02% of the ammonium sulfate solution.Based off of this information we can gather that the LC50 is at 0.01% ammonium sulfate concentration where the mortality of the Daphnia is 50%. Errors such as inaccurate measurements of the (NH₄)₂SO₄, cross contamination between the solutions (through graduated cylinders or pipets), or various uncontrollable conditions of the Daphnia magna (age, life expectancy, etc.) could have occurred during the procedure which caused the data to be altered. According to the data presented in this lab, the level of water toxicity that terminates half of the sample population is 0.01%, meaning that an ammonium sulfate solution of 0.01% is lethal to 50% of the Daphnia in the sample. This proves the hypothesis stating that "if Daphnia were to be exposed to various concentrations of ammonium sulfate, then the LC50 would be at a 0.03% concentration of ammonium sulfate" false. The data does not support this hypothesis as evidenced by the LC50 in the hypothesis being 0.03% ammonium sulfate solution and the actual LC50 of the data being 0.01%. Overall, this experiment has revealed that, at least for the Daphnia magna, any level of water toxicity higher than 0.01% is lethal to the population.
According to the results in Table 1 and Table 2, the Daphnia magna all die starting at the 0.02% concentration of (NH₄)₂SO₄ with the exception of only one death in the 0.0% concentration in the class data. The graph illustrates that the mortality of the Daphnia quickly rises from 0% to 100% in the concentrations between 0.0% and 0.02% of the ammonium sulfate solution.Based off of this information we can gather that the LC50 is at 0.01% ammonium sulfate concentration where the mortality of the Daphnia is 50%. Errors such as inaccurate measurements of the (NH₄)₂SO₄, cross contamination between the solutions (through graduated cylinders or pipets), or various uncontrollable conditions of the Daphnia magna (age, life expectancy, etc.) could have occurred during the procedure which caused the data to be altered. According to the data presented in this lab, the level of water toxicity that terminates half of the sample population is 0.01%, meaning that an ammonium sulfate solution of 0.01% is lethal to 50% of the Daphnia in the sample. This proves the hypothesis stating that "if Daphnia were to be exposed to various concentrations of ammonium sulfate, then the LC50 would be at a 0.03% concentration of ammonium sulfate" false. The data does not support this hypothesis as evidenced by the LC50 in the hypothesis being 0.03% ammonium sulfate solution and the actual LC50 of the data being 0.01%. Overall, this experiment has revealed that, at least for the Daphnia magna, any level of water toxicity higher than 0.01% is lethal to the population.